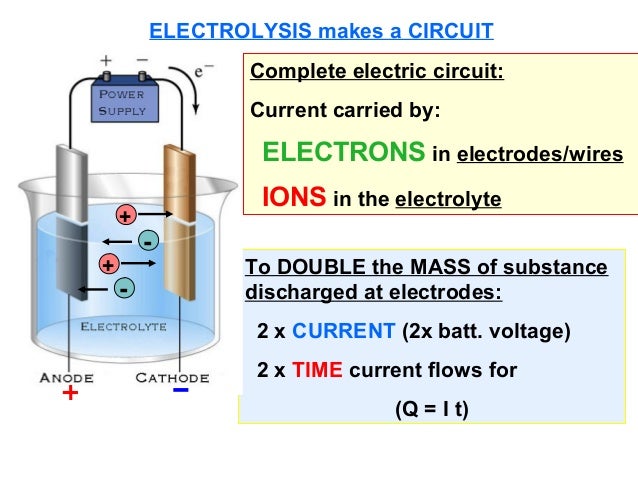
Hello, I have a question about electrolysis and how the metal moves. Everything I've read doesn't really answer the question outright but suggests that the metal moves from the pan to the sacrificial steel. However, some talk about the metal builds on the pan from the sacrificial steel. If the first statement is true, I can see how everything is cleaned from the pan but not why the sacrificial steel is named the way it is. Sacrifice usually means losing, not gaining. If the second statement is true, then that means all the carbon and rust is just plated over but the name then fits the sacrificial steel. :shootself: :???: :???: :headpop:
I'm one of those "need to know" types.
Thanks in advance :covri:
I'm one of those "need to know" types.
Thanks in advance :covri: